The modern society consumes much more energy and produces more environmental-hazard waste than any past age in the human history. To meet these challenges, our research is focused on the electrochemical energy conversion and environmental-related studies, aiming to promote the fundamental understanding of environmentally benign energy systems, such as solar cells and fuel cells, as well as the understanding of molecular adsorption and reaction mechanisms at electrode-electrolyte interfaces.
Our research interest is focused on the synthesis of nanomaterials and their applications in energy conversion. We fabricate energy conversion devices (photovoltaic or solar cells) using as-prepared nanomaterials and custom-designed dye molecules, and then investigate parameters that affect the device efficiency and charge transport mechanism. We are also interested in the charge transport in single molecule devices and molecular adsorption at electrode-electrolyte interfaces using electrochemical scanning probe microscopy (STM), atomic force microscopy (AFM), and electrochemical techniques. Specifically, a few projects are on-going or planned to investigate in the lab:
Preparation of semiconductor nanomaterials and applications in photovoltaic cells
Semiconductors have demonstrated great capacity for application in several fields, such as energy storage devices, solar light conversion, water-splitting and so on. The semiconductor to be utilized in these applications must hold appropriate physio-chemical properties including suitable band gap, proper mechanical and electrochemical resistance, and relatively stable excitation state among others. Promoting the desired properties could be achieved by structural doping of the materials with certain elements, substitution of rare and costly elements with less expensive ones, application of novel methods for limiting particle distribution and via fabrication of composite using carbon allotropes such as graphene. In this project, a variety of semiconductor nanomaterials will be prepared and these materials will be applied to photovoltaic cells for charge transport study. Their applications in the water-splitting and CO2 reduction will also be explored.
Our research interest is focused on the synthesis of nanomaterials and their applications in energy conversion. We fabricate energy conversion devices (photovoltaic or solar cells) using as-prepared nanomaterials and custom-designed dye molecules, and then investigate parameters that affect the device efficiency and charge transport mechanism. We are also interested in the charge transport in single molecule devices and molecular adsorption at electrode-electrolyte interfaces using electrochemical scanning probe microscopy (STM), atomic force microscopy (AFM), and electrochemical techniques. Specifically, a few projects are on-going or planned to investigate in the lab:
Preparation of semiconductor nanomaterials and applications in photovoltaic cells
Semiconductors have demonstrated great capacity for application in several fields, such as energy storage devices, solar light conversion, water-splitting and so on. The semiconductor to be utilized in these applications must hold appropriate physio-chemical properties including suitable band gap, proper mechanical and electrochemical resistance, and relatively stable excitation state among others. Promoting the desired properties could be achieved by structural doping of the materials with certain elements, substitution of rare and costly elements with less expensive ones, application of novel methods for limiting particle distribution and via fabrication of composite using carbon allotropes such as graphene. In this project, a variety of semiconductor nanomaterials will be prepared and these materials will be applied to photovoltaic cells for charge transport study. Their applications in the water-splitting and CO2 reduction will also be explored.
AFM images of synthesized NiWO4 Lab-prepared photovoltaic cells for
nano materials on mica surface energy-conversion study
nano materials on mica surface energy-conversion study
Electrochemical scanning tunneling microscopy (EC-STM) study of electro-catalytic oxidation of CO and CO2 reduction
Fuel cells, for example, direct methanol fuel cells (DMFCs) have many advantages over traditional batteries, such as portability and they are a clean (environment-friendly) energy resource, and are regarded as a possible future energy device. However, in the fuel cell reaction process, the reaction intermediate, carbon monoxide (CO), strongly binds to the catalyst surface (Pt) and blocks the active sites, so-called poisoning of the anodic catalyst. This poisoning of the catalyst by CO limits the performance of DMFC and challenges the development of platinum catalyst. Thus, studying CO adsorption at noble metal electrode/electrolyte is important from both fundamental and application aspects. Herein, we aim to use state-of-the–art EC-STM in combination with classic electrochemical techniques to explore the reaction mechanism at the molecular or atomic level. The study may provide valuable information and experimental guidance to develop high-performance anode catalysts, and effective electrode materials and facilitate the development of energy conversion devices.
Adsorption of molecules at electrode/electrolyte interfaces and single molecule charge transport in molecular junctions
The self-assembly of supramolecular structures is based on noncovalent interactions such as hydrogen-bond, van der Waals force, and metal ion-ligand coordination, etc. Both the molecule-molecule interaction (intermolecular) and molecule-substrate (often metal electrode) interaction play a crucial role in determining the structural motif of supramolecular architectures formed. Studying the interactions between adsorbates (molecules) and metal substrates can provide valuable information to other chemical reactions and processes such as organic phase transition and asymmetric heterogeneous catalysis. In this study, adsorption of aromatic hydrocarbon derivatives, including single ring and polycyclic hydrocarbons, at gold and platinum will be performed in electrochemical environment. Molecular assembly of aromatic hydrocarbon derivatives at solid-liquid interfaces and their charge transport properties at a single-molecule level will be studied by electrochemical STM and STM break junction techniques.
Fuel cells, for example, direct methanol fuel cells (DMFCs) have many advantages over traditional batteries, such as portability and they are a clean (environment-friendly) energy resource, and are regarded as a possible future energy device. However, in the fuel cell reaction process, the reaction intermediate, carbon monoxide (CO), strongly binds to the catalyst surface (Pt) and blocks the active sites, so-called poisoning of the anodic catalyst. This poisoning of the catalyst by CO limits the performance of DMFC and challenges the development of platinum catalyst. Thus, studying CO adsorption at noble metal electrode/electrolyte is important from both fundamental and application aspects. Herein, we aim to use state-of-the–art EC-STM in combination with classic electrochemical techniques to explore the reaction mechanism at the molecular or atomic level. The study may provide valuable information and experimental guidance to develop high-performance anode catalysts, and effective electrode materials and facilitate the development of energy conversion devices.
Adsorption of molecules at electrode/electrolyte interfaces and single molecule charge transport in molecular junctions
The self-assembly of supramolecular structures is based on noncovalent interactions such as hydrogen-bond, van der Waals force, and metal ion-ligand coordination, etc. Both the molecule-molecule interaction (intermolecular) and molecule-substrate (often metal electrode) interaction play a crucial role in determining the structural motif of supramolecular architectures formed. Studying the interactions between adsorbates (molecules) and metal substrates can provide valuable information to other chemical reactions and processes such as organic phase transition and asymmetric heterogeneous catalysis. In this study, adsorption of aromatic hydrocarbon derivatives, including single ring and polycyclic hydrocarbons, at gold and platinum will be performed in electrochemical environment. Molecular assembly of aromatic hydrocarbon derivatives at solid-liquid interfaces and their charge transport properties at a single-molecule level will be studied by electrochemical STM and STM break junction techniques.
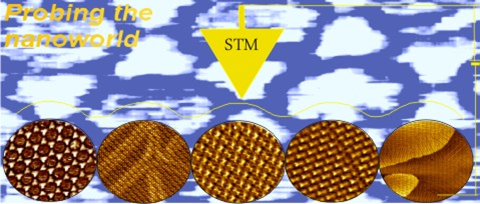
Five highly ordered nanostructures of trimesic acid molecules on Au(111) electrode have been fabricated and in-situ imaged by scanning tunneling microscopy techniques.